Filed by Mirna Therapeutics, Inc. Pursuant to Rule 425 under Securities Act of 1933, as amended and deemed filed pursuant to Rule 14a-12 under the Securities Exchange Act of 1934, as amended Subject Company: Mirna Therapeutics, Inc.
Subject Company’s Commission File No.: 001-37566 Date: June 9, 2017

Synlogic Corporate Overview JC Gutierrez-Ramos, President and CEO June 2017 A Novel Class of Living Medicines Synthetic BioticTM medicines to perform metabolic functions to treat diseases throughout the body

Forward Looking Statements This presentation contains “forward-looking statements” that involve substantial risks and uncertainties for purposes of the safe harbor provided by the Private Securities Litigation Reform Act of 1995. All statements, other than statements of historical facts, included in this presentation regarding strategy, future operations, future financial position, future revenue, projected expenses, prospects, plans and objectives of management are forward-looking statements. In addition, when or if used in this presentation, the words “may,” “could,” “should,” “anticipate,” “believe,” “estimate,” “expect,” “intend,” “plan,” “predict” and similar expressions and their variants, as they relate to Mirna, Synlogic or the management of either company, before or after the proposed merger, may identify forward-looking statements. Examples of forward-looking statements include, but are not limited to, statements relating to the timing and completion of the proposed merger; Mirna’s continued listing on the NASDAQ Global Market until closing of the proposed merger; the combined company’s listing on the NASDAQ Global Market after closing of the proposed merger; expectations regarding the capitalization, resources and ownership structure of the combined company; the approach Synlogic is taking to discover and develop novel therapeutics using synthetic biology; the adequacy of the combined company’s capital to support its future operations and its ability to successfully initiate and complete clinical trials; the nature, strategy and focus of the combined company; the difficulty in predicting the time and cost of development of Synlogic’s product candidates; the executive and board structure of the combined company; and expectations regarding voting by Mirna’s and Synlogic’s stockholders. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors, including, without limitation: the risk that the conditions to the closing of the transaction are not satisfied, including the failure to timely or at all obtain stockholder approval for the transaction; uncertainties as to the timing of the consummation of the transaction and the ability of each of Mirna and Synlogic to consummate the transaction; risks related to Mirna’s ability to correctly estimate its operating expenses and its expenses associated with the transaction; the ability of Mirna or Synlogic to protect their respective intellectual property rights; unexpected costs, charges or expenses resulting from the transaction; potential adverse reactions or changes to business relationships resulting from the announcement or completion of the transaction; and legislative, regulatory, political and economic developments. The foregoing review of important factors that could cause actual events to differ from expectations should not be construed as exhaustive and should be read in conjunction with statements that are included herein and elsewhere, including the risk factors included in Mirna’s Quarterly Report on Form 10-Q filed with the SEC on May 9, 2017. Mirna and Synlogic can give no assurance that the conditions to the transaction will be satisfied. Except as required by applicable law, Mirna and Synlogic undertake no obligation to revise or update any forward-looking statement, or to make any other forward-looking statements, whether as a result of new information, future events or otherwise.

Additional Information This presentation is not intended to and does not constitute an offer to sell or the solicitation of an offer to subscribe for or buy or an invitation to purchase or subscribe for any securities or the solicitation of any vote in any jurisdiction pursuant to the proposed transaction or otherwise, nor shall there be any sale, issuance or transfer of securities in any jurisdiction in contravention of applicable law. No offer of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the United States Securities Act of 1933, as amended. Subject to certain exceptions to be approved by the relevant regulators or certain facts to be ascertained, the public offer will not be made directly or indirectly, in or into any jurisdiction where to do so would constitute a violation of the laws of such jurisdiction, or by use of the mails or by any means or instrumentality (including without limitation, facsimile transmission, telephone and the internet) of interstate or foreign commerce, or any facility of a national securities exchange, of any such jurisdiction. In connection with the proposed transaction between Mirna and Synlogic, Mirna intends to file relevant materials with the SEC, including a registration statement that will contain a proxy statement and prospectus. MIRNA AND SYNLOGIC URGE INVESTORS AND STOCKHOLDERS TO READ THESE MATERIALS CAREFULLY AND IN THEIR ENTIRETY WHEN THEY BECOME AVAILABLE BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT MIRNA, SYNLOGIC, THE PROPOSED TRANSACTION AND RELATED MATTERS. Investors and shareholders will be able to obtain free copies of the proxy statement, prospectus and other documents filed by Mirna with the SEC (when they become available) through the website maintained by the SEC at www.sec.gov. In addition, investors and stockholders will be able to obtain free copies of the proxy statement, prospectus and other documents filed by Mirna with the SEC by contacting Investor Relations by mail at Attn: Investor Relations, PO Box 163387, Austin, TX 78716. Investors and stockholders are urged to read the proxy statement, prospectus and the other relevant materials when they become available before making any voting or investment decision with respect to the proposed transaction. Mirna and Synlogic, and each of their respective directors and executive officers and certain of their other members of management and employees, may be deemed to be participants in the solicitation of proxies in connection with the proposed transaction. Information about Mirna’s directors and executive officers is included in Mirna’s Annual Report on Form 10-K for the year ended December 31, 2016, filed with the SEC on March 15, 2017. Additional information regarding these persons and their interests in the transaction will be included in the proxy statement relating to the transaction when it is filed with the SEC. These documents can be obtained free of charge from the sources indicated above.
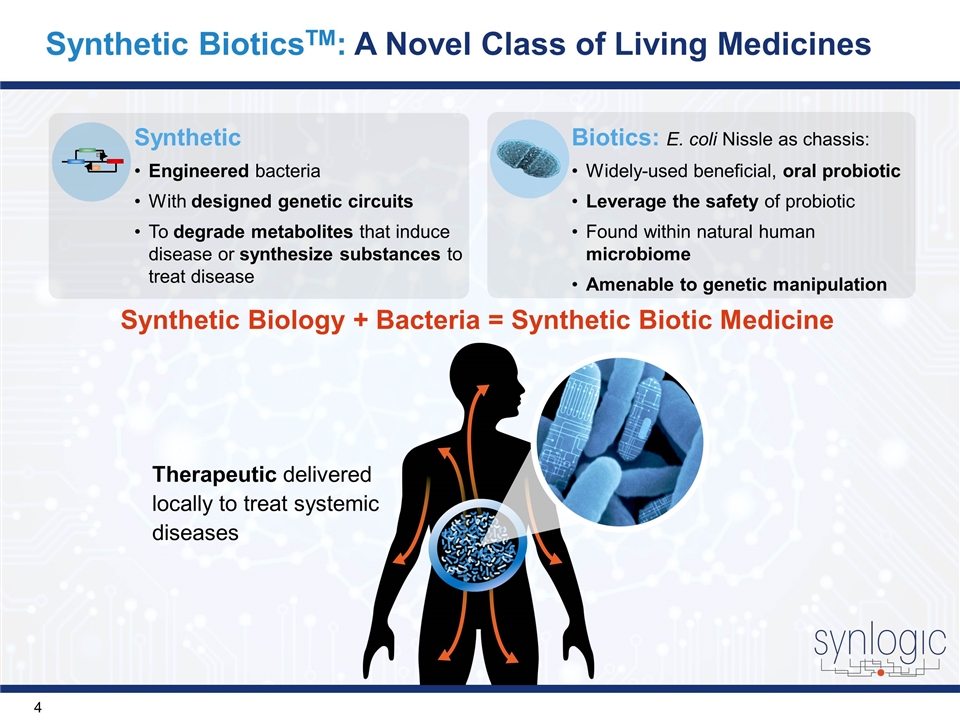
Synthetic BioticsTM: A Novel Class of Living Medicines Synthetic Biology + Bacteria = Synthetic Biotic Medicine Therapeutic delivered locally to treat systemic diseases Synthetic Engineered bacteria With designed genetic circuits To degrade metabolites that induce disease or synthesize substances to treat disease Biotics: E. coli Nissle as chassis: Widely-used beneficial, oral probiotic Leverage the safety of probiotic Found within natural human microbiome Amenable to genetic manipulation

Synlogic announced merger with Mirna Therapeutics to become Nasdaq listed company The go forward company will operate as Synlogic, Inc. Expected to close 3Q 2017 subject to the approval of the stockholders of each company and the satisfaction or waiver of other customary conditions Expected ownership split subject to adjustment based on Mirna’s net cash at closing Synlogic Shareholders: Approximately 83% Mirna Shareholders: Approximately 17% Synlogic funding expected to support operations through mid-2019. Includes: $42M from Series C Approximately $40M cash expected from merger at time of close Synlogic existing cash at time of close Transaction Overview

Synlogic Management Team: From Funding of Platform to Clinic in Less than Three Years JC Gutierrez-Ramos, CEO Group SVP Biotherapeutics, Pfizer SVP, Head Immunoinflammation Center for Drug Discovery, GSK CSO & Site Head, Amgen Mountain View Dean Falb, CTO Entrepreneur in Residence, Atlas Venture VP, R&D, Stryker Regenerative Medicine Aoife Brennan, CMO VP, Rare Disease Innovation Unit, Biogen Medical Director, Tolerx Caroline Kurtz, Translational Science Vice President, GCC Platform Lead, Ironwood Pharmaceuticals Director, Infectious Diseases, Genzyme Todd Shegog, CFO SVP & CFO, Forum Pharmaceuticals SVP & CFO, Millennium Pharmaceuticals Dick Schwartz, SVP, Manufacturing Chief, Vaccine Production Program Lab, NIH Senior Director, Process & Manufacturing Sciences, MedImmune Paul Miller, CSO VP, Infection iScience, AstraZeneca VP, Antibacterials Research Unit, Pfizer

Synthetic Biotic Broad Platform and Pipeline Potential: Focus on Orphan Metabolic Diseases Immuno-Oncology (IO) Synthetic Biotic Platform Internal Pipeline Focus Immunomodulation Inflammatory Bowel Disease On-going Partnership Other Inflammation and Immunology Metabolic Diseases Rare Diseases Inborn Errors of Metabolism Urea Cycle Disorder Phenylketonuria Organic Acidemias Branched Chain Amino Acid Disorders Hepatic Encephalopathy Obesity Type 2 Diabetes NASH Other Liver Diseases

Synthetic Biotic Pipeline Inborn Errors of Metabolism Metabolic Disease Immunomodulation Immuno Oncology 1 Indication Screening Lead Discovery Lead Optimization IND-Enabling Studies Phase I Hyperammonemia -Urea Cycle Disorder SYNB1020 Phenylketonuria SYNB1618 Organic Acidemias Maple Syrup Urine Disease Inflammatory Bowel Disease Immuno Oncology 2 Hyperammonemia Liver (HE) Immuno Oncology 3 SYNB1020

Synlogic’s Initial Focus: Inborn Errors of Metabolism Disease Toxic Metabolite Urea Cycle Disorders Ammonia Phenylketonuria Phenylalanine Organic Acidemias Propionate Maple Syrup Urine Disease Ile, Leu, Val Clinical Candidates Lead Optimization ~25K cases in the US

SYNB1020 for Hyperammonemia Indications: Urea Cycle Disorders (UCD) and Hepatic Encephalopathy (HE) H H H N Protein Metabolism (food, muscles, etc.) Ammonia Buildup (toxic levels) Urea (removed from the body normally) Ammonia Liver Healthy ! Hepatic Encephalopathy: Sub-population of cirrhosis patients Symptoms: Neurological dysfunction that develops in association with liver disease: cognitive, intellectual, neuromuscular, emotional Episodes lead to hospitalization Rifaximin approved in 2010 for reduction in risk of overt HE recurrence Urea Cycle Disorders: 2,000-3,000 patients with hereditary disorder (U.S.) Genetic defects in Urea Cycle Symptoms: vomiting, encephalopathy, respiratory stress, irreversible brain damage, coma, death Standard of care inadequate in that ammonia levels are not normalized and strict diet required. Best option is liver transplant Ravicti is a nitrogen-binding agent approved in 2013 Hyper-ammonemia

Probiotic bacteria: E. coli Nissle SYNB1020: Conversion of Toxic Ammonia into Beneficial Arginine for the Treatment of UCD and HE Synthetic Biotic Medicine Correct the deficiency ✓ UCD and HE Patients Target Profile: Normalize Ammonia Levels Dietary Protein Liberalization Without Systemic Toxicity Arginine argE argC argB argH R FNR FNR carA carB argG argl argD fbr-argA Ammonia Metabolic Conversions Arginine Synthetic Genetic Circuit Glutamate

Efficient Ammonia Conversion: In Vitro and In Vivo Blood Ammonia Reduced in vivo (spf-ash hyperammonia/UCD model on High Protein Diet) 0 100 50 150 200 250 Blood Ammonia (uM) Water/NC Water/HP SYNB1010/HP *** NC – Normal chow, HP – High protein chow SYNB1010 – arg producing, Thy A auxotrophy, Kan resistant (a kanamycin resistant version of SYNB1020 clinical candidate) *** Statistically significant Ammonia to Arginine Conversion in vitro Nissle – E. coli Nissle 1917 Strep resistant control strain SYNB1010–arginine producing, Thy A auxotrophy, Kan resistant SYNB1020-arginine producing, Thy A auxotrophy, clinical candidate Mean Arg production rate=650nmoles/109 cells/hr Arginine Production (nmoles/109cells) Time (min) 20 40 60 80 100 120 250 500 750 1000 1250 1500 Nissle Control SYNB1010 SYNB1020

SYNB1020: Toxicology Package and Regulatory Path Regulatory Orphan Drug Designation Pre-IND Meeting Feedback from FDA Office of Vaccines Research and Review No Recombinant DNA Advisory Committee (RAC) CMC plan for the Phase 1/2 studies Non-Clinical plan: dose selection; single species tox; single auxotrophy Alignment with Phase 1 clinical plan: SAD/MAD in healthy volunteer Lowering of blood ammonia level is an approvable end-point Safety Toxicology Toxicology program completed Clean safety profile No toxicity at highest feasible dose in two species No evidence of distribution outside the GI tract

From Flask to Industrial Fermenter at CMO: Well-Controlled Process at 150L Successful Scale-up to Biomass for Clinical Scale Number of doses at 1e12 CFU 10000 1000 100 10 Shake flask – 250 mL Shake flask – 500 mL Shake flask – 1 L Bench fermenter – 5 L Pilot fermenter – 25 L Pilot fermenter – 150 L Engineering Run N=2 – 150 L Pilot fermenter – 15 L 6 12 24 100 300 500 3000 3000 Increasing Time Post Induction Run 1 0hr-PI Run 1 1hr-PI Run 1 2hr-PI Run 1 3hr-PI Ref Std. 1.25 1.00 0.75 0.50 0.25 Arginine (umol/hr/1e9 cells) Control of Activity During Manufacturing Hours Post Induction Clinical Trial Run – 150 L 3000

Estimated start mid-2017 Traditional SAD/MAD design assessing safety, tolerability and kinetics in healthy volunteers across a range of doses Includes exploratory biomarkers in the context of controlled calorie and protein diet Complete urine and fecal collection Blood ammonia and amino acid profile analysis SYNB1020: Phase 1 SAD/MAD in Healthy Volunteers Estimated Timing 2017 2018 SAD/MAD

SYNB1020 Strategy in Hyperammonemia: Following Success in Healthy Volunteers, Two Patient Studies Planned Phase 2 UCD and/or HE Phase 1b UCD Goal: Demonstrate safety/tolerability in UCD patients Phase 1b/2a Cirrhosis with Elevated Ammonia Goal: Establish safety/tolerability and POC in patients (reduction of ammonia) Phase 1 HV SAD/MAD Goal: Establish SYNB1020 safety, tolerability, kinetics across range of doses. Exploratory Biomarkers

SYNB1618: Phenylketonuria (PKU) Overview Protein Metabolism (food, muscles, etc.) Phenylalanine Buildup (toxic levels) Tyrosine (used by body normally) Phenylalanine Liver Healthy PKU ! PKU disorders: 16,500 patients with hereditary disorder (U.S.) Genetic defect in phenylalanine hydrolase (PAH) enzyme Symptoms: mental retardation, convulsions, behavior problems, skin rash, musty body odor Kids maintained on very low protein diet (NO meat, dairy, dry beans, nuts, eggs) Kuvan, a cofactor for PAH enzyme, launched in 2007 for a segment of the patient population

SYNB1618: Degradation of Toxic Phenylalanine for the Treatment of PKU Probiotic bacteria: E. coli Nissle Synthetic Biotic Medicine Correct the deficiency ✓ Phenylalanine t-Cinnamic Acid Synthetic Genetic Circuit Phenylalanine t-Cinnamic Acid Metabolic Conversions High-Affinity Uptake LAAD Phenylketonuria Patients Hippurate FNR FNR PAL3 FNR FNR pheP AraC AraC FNR Target Profile: Oral Normalize Phenylalanine Levels Dietary Liberalization Without Systemic Toxicity

PKU: Efficient Phe Degradation In Vitro and In Vivo With pheP Transporter Without pheP Transporter 20 10 10 0 5 Phe Degradation Rate (μmoles/h/10e9cells) Integration Site PAL copy Number Integration Site PAL copy Number pheP copy Number LAAD + 3XPAL + pheP+ PAL – Convert Phe to TCA pheP – Transport Phe into cell LAAD – Convert Phe to PPA Lead Optimization Based on Phe Degradation in vitro Greater Consumption Less Consumption Blood Phe is Blunted in Activity Dependent Manner in vivo PKU Model

PKU: Urinary Hippurate as a Biomarker for Synthetic Biotic Program Activity Urinary Hippurate (umol), 0-6 h Increased Urinary Hippurate in NHP Following Treatment with Synthetic Biotic Program *Average quantity of hippurate recovered per cage Increased Urinary Hippurate in Mice Following Treatment with Synthetic Biotic Program– Dose Response with Clinical Candidate

SYNB1618: Clinical Candidate for PKU Traditional SAD/MAD design assessing safety, tolerability and kinetics in healthy volunteers (HV) across a range of doses Includes a cohort of 8 PKU patients Proof of Mechanism: Hippurate production in HV Will assess reduction in plasma phenylalanine SYNB1618: Planning Phase 1 SAD/MAD in Healthy Volunteers with Patient Cohort Patient Cohort SAD/MAD 2017 2018

Key Inflection Points and Estimated Timing: SYNB1020 and SYNB1618 SYNB1020 Estimated Timing: Mid-2017: HV: Safety, tolerability, kinetics First half of 2018: Cirrhosis patients with elevated ammonia initiates: Safety/tolerability and POC in patients (reduction of ammonia) Mid 2018: UCD patient cohort initiates: Safety, tolerability SYNB1618 Estimated Timing: First half of 2018: HV SAD/MAD, then initiate patient cohort Evaluate Proof of Mechanism: Hippurate production Evaluate Proof of Concept: Reduction of phenylalanine SYNB1020 Hyperammonemia SYNB1618 PKU 22 P1b/2a Cirrhosis Patients with Elevated Ammonia P1b UCD Safety/Tolerability SAD/MAD HV PKU Cohort Healthy Volunteer SAD/MAD

Synthetic Biotic Medicines: Applicability Beyond Rare Disease Across Multiple Pathways in Metabolic & Autoimmune Diseases and Immuno Oncology Short Chain Fatty Acids: Butyrate Acetate Propionate Proteins and Peptides IL2, IL12, IL15, IL22,IL10 IFN, TNF, CXCL10, CD40L, hyaluronidase GLP1, GLP2 Amino Acid Metabolism Branched Chain AAs Trp, Kyn, indoles, Serotonin, Melatonin Arginine Phenylalanine Multiple MoAs in ONE Synthetic Biotic Combination therapy in a single NME scFv Display Anti-PD1 Anti-CD47 Degradation of Toxic Metabolites: Ammonia Nucleotides 14 Issued/Allowed Patents 41 Patent Families 165 Patent Applications* Inflammation/ Metabolic Metabolic Metabolic Inflammation Immuno Oncology Inflammation Immuno Oncology Immuno Oncology * As of May 2017


Synlogic Overview Novel Therapeutic Class Robust Pipeline with Orphan Drug Programs Broad Platform - Multiple Product Opportunities Dominant Synthetic Biotic IP Portfolio As of May 2017 Strong Balance Sheet Synthetic Biotic Platform: Leading in the use of synthetic biology to genetically reprogram beneficial probiotics to have transformative impact on treatment of human disease Simple, robust and rapid process for the creation of drug candidates Founded out of labs of James Collins and Timothy Lu at MIT SYNB1020 for Hyperammonemia including Urea Cycle Disorder (UCD) & Hepatic Encephalopathy (HE). Healthy volunteer study planned mid-2017 SYNB1618 for Phenylketonuria (PKU): Planning IND Raised ~$112M in three private rounds Investors include: Aju IB Investment, Ally Bridge Group, Atlas Venture, Deerfield Management, New Enterprise Associates (NEA), OrbiMed, Perceptive Advisors, Rock Springs Capital Highly Experienced Management Team JC Gutierrez-Ramos, CEO Aoife Brennan, CMO Todd Shegog, CFO Paul Miller, CSO Dean Falb, CTO Richard Schwartz, SVP Manufacturing Caroline Kurtz, VP Translational Science AbbVie: Inflammatory Bowel Disease (IBD) partnership Broad Pipeline with opportunities to partner in major indications- Immuno Oncology, Liver and Metabolic Diseases, Inflammation 14 Issued/Allowed Patents 40 Patent Families 165 Pending Patent Applications